Back to Index
ION SELECTIVE SCREEN (ISS): Research Notes
More than half a century ago when lead-acid battery manufacturers switched from wood veneer separators to synthetic materials, the changeover inadvertently caused battery life to crash. While the solution was found in the use of ligno-sulfonates, other than empirically re-formulating these substances, it seems hardly any investment has been made in the utilization of organic materials to purposefully continue to enhance battery performance. Ion Selective Screen (ISS) is designed to put organic technology back in the running so as to better equip the lead-acid battery industry to meet the inevitable difficult commercial challenges that lie ahead.
ISS can help to achieve calcium-like low water consumption from antimony alloy batteries - but to do so a formation catalyst in the electrolyte has to be used.
New antimony alloy positives give off a surge of antimony during formation that is deposited immediately on the negatives, causing the characteristic high water consumption of antimony alloy batteries. This is likely to be masked if the negatives also contain antimony in the grids but shows up clearly with calcium alloy negatives.
The catalyst has a half-life roughly equal to the duration of the formation charge - disappearing without a trace after only a few days.
The advantage of ISS plus formation catalyst are obvious. The use of recycled lead carrying a large impurity burden is facilitated. Batteries can be provided with long lasting antimony alloy positives and low water consuming calcium alloy negatives - resulting in the best of both worlds in service.
For example, the way would be cleared for industrial motive power batteries and golf cart batteries to be watered no more than once a year (!). Hybrid and electric automobile batteries could be made very inexpensively, yet provide a highly satisfactory battery life.
The following procedures are typical of 2-cell testing used to evaluate ISS. Each cell has a single positive and a single negative plate. The test cell has an antimony alloy positive, the control cell a calcium alloy positive and both cells have calcium alloy negative plates.
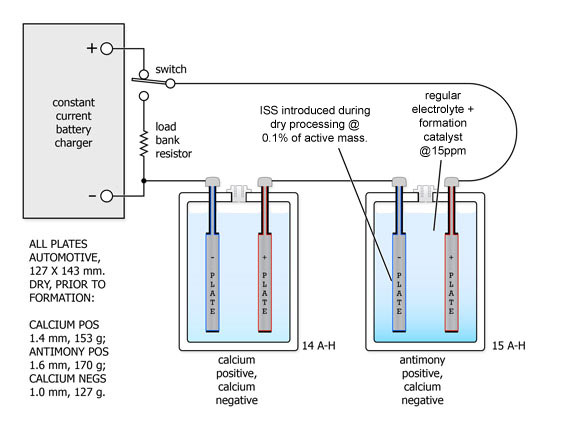
The two cells received formation charging connected in series and were subsequently deep cycled twice to ascertain their ampere-hour capacities. The antimony positive cell appeared to have about 7% more capacity than the calcium positive cell - likely due to its positive plate being fractionally thicker. The antimony's end-of-charge dV/dt had been considerably speeded up and more sharply defined.

The cells were reconnected as shown above and the potentials across both cells were carefully adjusted to match each other using a high resolution Hewlett-Packard digital voltmeter. The currents via the cells were measured with a specially constructed differential shunt resistor network having a matching accuracy batter than 0.1% . The current of the Ca-Ca cell was measured across A - B, the current of the Sb-Ca cell was measured across C - D and the differential was monitored across B - D, again using the H-P instrument.
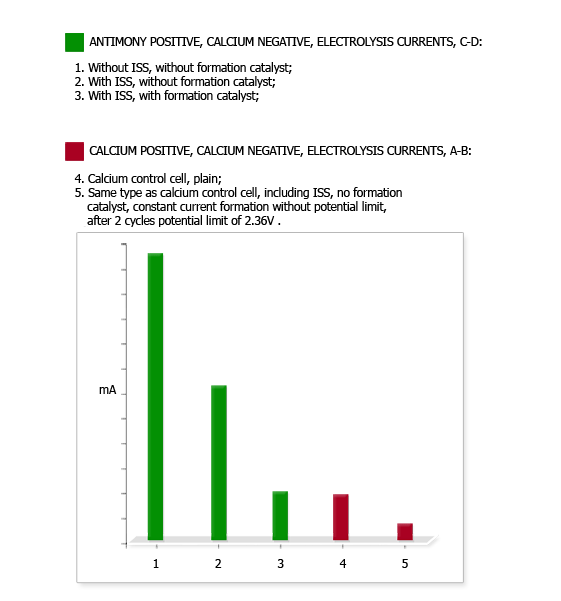
(In previous testing an untreated antimony cell had drawn more than six times the electrolysis current of a calcium cell. An antimony cell containing ISS in the negative but no formation catalyst in the electrolyte drew roughly three times the electrolysis current of the calcium cell.)
Subsequent cycling of the cells in parallel was performed. Discharging at the 5 hour rate down to 1.50 V/cell and charging at the 20 hour rate up to an average of 2.60 V/cell - holding this voltage for 5 hours and then lowering it to 2.36 V/cell for 24 hours to measure the electrolysis current.
After 10 cycles the electrolysis current of the Ca-Ca cell had fallen from 4.7 mA down 1.8 mA, while that of the Sb-Ca cell had remained at 5.2 mA. While implying an improvement in the Ca-Ca cell, individual cell capacity testing showed the Ca-Ca cell capacity had fallen from 14 A-H to 5 A-H, while the Sb-Ca cell capacity had risen slightly from 15 A-H to 17 A-H.
The positive plate of the Ca-Ca cell was replaced with an identical freshly formed and precycled plate, resulting in the electrolysis current rising to 4.6 mA and the capacity being restored to 14 A-H.
Had the Ca-Ca cell been defective? To find out, two more Ca-Ca cells were built, formed and tested in the same way. Both suffered similar capacity loss. Indeed, numerous publications have described this kind of capacity loss and therefore it must be accepted that lead-calcium alloy technology might well prove to be an evolutionary dead-end.
Since treatment assists in increasing battery life, the upsurge in water consumption at the end of the useful service life of a motive power battery is delayed. In this context battery leasing, once a year maintenance, at a fixed price - no watering hassles in between - seems to look set to become a reality.

|

